Water Drawdown Efficacy and Implications
Executive Summary
Macrophytes or Submerged Aquatic Vegetation (SAV) are often part of many aquatic systems. Carried by wind or waterfowl or (un)intentionally introduced by humans, SAV can quickly establish. Understanding the role that these plants play in the ecology of a system is an important step to developing methods to control their unwanted spread. Here, an overview is provided of the current SAV status of Deep Creek Lake, Maryland as well as an evaluation (literature review) of the potential effects of various control strategies (e.g., winter drawdowns) within the ecosystem, both biotic and abiotic.
Introduction
Lakes and reservoirs are frequently developed into recreational havens for boaters and fisherman alike. In addition, many serve concurrently in more industrial or ecological roles as hydroelectric power sources or as flood control/management devices. Conditions among many of these lentic systems often lend themselves to invasions of aquatic nuisance species like Zebra Mussels (Dreissena polymorpha) and Hydrilla (Hydrilla verticillata). Recreational boaters for example can easily introduce and spread such invaders unintentionally with movement of their vessels from waterbody to waterbody. In some instances, systems become overrun by these nuisance species which necessitates drastic measures be employed. Macrophytes or submerged aquatic vegetation (SAV), if left unchecked, can create severe problems, turning once productive systems into eutrophic nightmares.
Submerged aquatic vegetation is defined as plants that are usually rooted in the bottom soil with the vegetative parts predominantly submerged. While rooted vegetation can provide shelter, habitat, and food for many organisms as well as oxygen through the process of photosynthesis, some plants pose threats to aquatic systems choking out native species. This review serves as an assessment of the effects of winter drawdowns, both positive and negative across various taxa. Other control methods are
discussed, but the focus will be water withdrawal (mechanical control). It is intended to provide guidance for the Deep Creek Watershed Foundation as they tackle increases in SAV around the Lake.
Current Status of SAV in Deep Creek
Vegetation surveys were performed by MD Department of Natural Resources (DNR) between 2018 and 2020 to track SAV movement and expansion in Deep Creek Lake (Table 1). Three species appear to dominate: a species complex of Water Milfoil (Myriophyllum spp.), Broad-leafed Pondweed (Potamogeton amplifoilus), and Wild Celery or Tape Grass (Vallisneria americana). For the purposes of discussion, these three macrophytes will be the focus to assess the varying ecological effects of SAV.
Table 1 – Summary of vegetation present in Deep Creek*. Data collected by and obtained from the MD Department of Natural Resources. I = introduced and N = native.
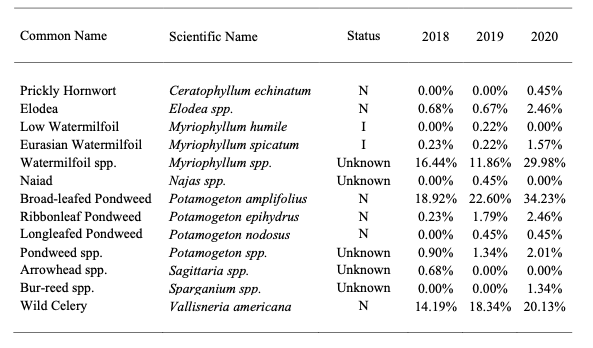
A. Water Milfoil Complex and Eurasian Watermilfoil
There are native species of milfoil that may benefit fish by providing cover and by serving as a source of food for various wildlife species; however, its prolific growth often limits these values. In fact, excessive growth can reduce oxygen supplies that result in fish kills. Perhaps the most concerning species of milfoil is the Eurasian Watermilfoil (Myriophyllum spicatum, EWM), a native to Europe, Asia and North Africa (Nichols and Shaw 1986; Eiswerth et al. 2000; Fig. 1), and one that, while identified by the MD DNR, is likely a component of the mifloil complex (Table 1).

Figure 1 – Image showing native Low Watermilfoil (left) and introduced Eurasian Watermilfoil (right). While the exact dates of its introduction in the United States are unknown, it likely occurred
sometime after the 1880s. It is often confused with the native Northern Milfoil (Myriophyllum exalbescens) and although its density in Deep Creek Lake appears low for now, it has the ability to rapidly spread.
Ecologically, EWM presents some issues due to its prolific reproduction (it can reproduce both sexually and asexually) and ability to crowd out native species (Smith and Barko 1990). In addition, EWM spreads when brittle plant pieces break off, float on the water currents, and re-root elsewhere (Aiken et al. 1979; Madsen et al. 1988). It may also hitchhike on boats and fishing gear, increasing its
movement within a lake or between neighboring bodies of water (Johnstone et al. 1985). It is typically most abundant in water between 1 and 4 m deep (Nichols and Shaw 1986) but can be found at depths of up to 7 m (Reznicek et al. 2011) or as deep as 10 m (Aiken et al. 1979). When it reaches the water’s surface it can form a dense canopy mat (Aiken et al. 1979).
Invertebrate and fish communities in EWM differ from those associated with other SAV. Dvorak and Best (1982) found that EWM had the poorest macroinvertebrate fauna and 3 to 4 times fewer fish than pondweed-wild celery communities. The characteristics described above make the milfoil complex a group to watch vigilantly, curbing its spread to reduce its chances of forming dense aggregations.
B. Broad-leafed Pondweed
A North American native, Broad-leafed Pondweed (BLP; Potamogeton amplifolius) has increased in size within Deep Creek, becoming a species dominant (Table 1; Fig. 2). Pondweeds can provide excellent fish cover (Craig 1952). BLP is often found at depths between 1 and 4 m (Wakeman and Les 1994) in calm water which is preferred by Bluegill (Lepomis macrochirus) for nesting and feeding (Tomcko and Pereira 2006).

However, if left unchecked BLP can grow pervasively, dispersing quickly within an aquatic system. Options available to curb its spread include biological, chemical, or mechanical removal (Texas A&M 2022b). Biological removal would require introducing Grass Carp (Ctenopharyngodon idella); but this would not be an option in Maryland as the Department of Natural Resources has banned this fish from all waterways (MICRA 2015). Chemical removal using various herbicides (Texas A&M 2022b) has also been proposed as a removal method but may be viewed negatively by the public. Mechanical removal by raking or dredging the waterbody is an option likely not available to Deep Creek Lake due to the recreational activities that are a focus of this waterbody. However, winter drawdowns (more on this below), another mechanical control, may be a more viable option, especially in large lake or reservoir systems. This will be discussed further below.
C. Wild Celery
Wild Celery (WC, Vallisneria americana) is a fast-growing perennial aquatic macrophyte (Fig. 3). With ribbon like leaves it is often found rooted in mud, growing among pondweeds. In lakes it can grow to a few meters in length and, if the substrate is suitable, form large mats of grass in late summer (Korschgen and Green 1988). It provides shelter for juvenile fish like Perch (Perca flavescens) and provides cover for Bass (Micropterus sp.) and Bluegill. It is also an excellent food choice for waterfowl, particularly diving ducks, like Canvasbacks (Aythya valisineria) and Scaups (Aythya sp.) (Korschgen and Green 1988).
This plant is best left alone because of the benefits it offers to fish and wildlife species; however, if left unchecked it may interfere with recreational activities like boating and fishing. Large aggregations can foul outboard motors and the thin ribbon-like leaves can become attached to fishing lures. Herbicides do not work well to control this plant and mechanical removal often fails because uprooted plants can re-establish themselves (MN DNR 2022; Texas A&M 2022a).

Summary of SAV in Deep Creek and proposed management strategy
The aforementioned section describes the general nature of three-dominant plants (complexes) present in Deep Creek Lake, each with their own benefits and dispersal strategies. While some are viewed as nuisance species others are viewed as very beneficial to fish and wildlife alike. Management of SAV is paramount to the continuing success of Deep Creek Lake. WDFW (2015) recommends developing a vegetation management plan to control and remove aquatic plants. A vegetation management plan is a comprehensive approach to controlling aquatic plants where all strategies are first considered and usually some combination of techniques is selected and implemented. These plans should be based on the biology and ecology of the aquatic plant(s) to be controlled and the environmental characteristics of the site. As a result, data (e.g., Table 1) about the system should first be assembled and analyzed.
Of the three basic control methods (mechanical, biological, and chemical), mechanical control can be the least expensive and most convenient. Moreover, public support is often greater for this method because chemicals are not being introduced into the environment (University of Florida 2021),
or a non-native species like Grass Carp. Deep Creek Watershed Foundation is proposing to employ a seasonal drawdown strategy to control the spread of several aquatic macrophytes.
Unlike raking or physical removal, winter drawdown exposes plants to air-drying and freezing temperatures. The desiccation process can accelerate erosional processes which reduce fine-textured sediment (Effler and Matthews 2004; Cooley and Franzin 2008), organic matter, and nutrients (James et al. 2001; Furey et al. 2004). This in turn can leave behind larger sediment particles with low nutrient storage capacity. These abiotic changes along with direct physiological stresses from desiccation and freezing conditions can reduce macrophyte abundance and alter assemblage composition (Wilcox and Meeker 1991; Wagner and Falter 2002; Turner et al. 2005; Carmignani and Roy 2017).
The practice of water level manipulation reduces water levels to some predetermined quantity and is generally designed to expose portions of a lake bottom inundated with nuisance plants. Manipulating water levels (drawdowns) to remove or control aquatic noxious weeds and/or exposing plants and root systems to extreme temperature and moisture conditions may be appropriate under certain circumstances (WDFW 2015). Winter drawdowns and subsequent spring refills (Fig. 4) are a common lake and reservoir management practice to achieve a variety of human goals.
Drawdowns occur as early as November/December and can continue through April (NYSDEC 2005, Carmignani and Roy 2017; Fig. 4). However, this timetable can vary depending on the management objective and the seasonal climate (WDFW 2015). Historically, fishery managers used winter drawdowns to concentrate prey populations, this in turn stimulated piscivorous sport fishes by reducing prey refuge habitat (Hulsey 1957; Lantz et al. 1967; Groen and Schroeder 1978). This action promoted macrophyte growth which served as important spawning and rearing areas for these species (Fox et al. 1977). Fisheries managers also used drawdowns to attempt to eradicate undesired fishes
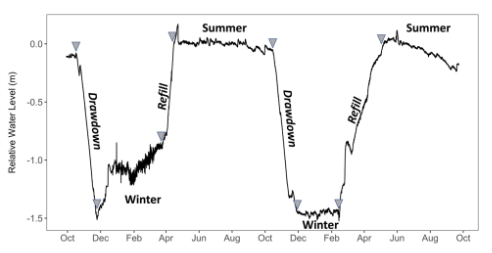
(Verrill and Berry Jr. 1995). Today, drawdowns have expanded value because many reservoirs have been developed for hydroelectric power generation. Winter drawdowns can help to meet the increased energy demand in such reservoirs during winter months (Aroviita and Hämäläinen 2008) and provide storage in anticipation of seasonal spring flooding (Hellsten 1997).
In recreational lakes, annual winter drawdowns can serve as a preventative measure to protect docks and retaining walls from ice scour damage, permit shoreline cleanup, and reduce nuisance levels of aquatic vegetation (Cooke et al. 2005). This can be an effective control technique, especially if done in successive years (NYSDEC 2005; Carmignani and Roy 2017). Water level manipulation can also provide a good opportunity to perform minor dam repairs and improve or enhance shorelines. Here, a literature review is provided that explores the ecosystem implications (both biotic and abiotic) of winter drawdowns.
A. Plants/Habitat
Studies have documented reduced macrophyte biomass and density from drawdown exposure compared to reference systems or previous non-drawdown conditions (Tarver 1980; Wagner and Falter 2002; Turner et al. 2005; Beklioglu et al. 2006; Sutela et al. 2013). Drawdowns of relatively large amplitude (e.g., >2-3 m) can significantly reduce macrophytes (Rorslett 1989; Turner et al. 2005; Keto et al. 2006; Sutela et al. 2013), while relatively mild drawdowns (<1.5 m) show less impact on density (Keto et al. 2006), potentially increasing macrophyte richness (Mjelde et al. 2012).
Response to drawdowns can be species specific and in some cases nuisance species are stimulated to grow (Cooke 1980). Turner et al. (2005) monitored the aquatic plant community in an experimental lake drawdown. They documented an initial two-thirds reduction in littoral macrophytes, however, recovery was variable among species. Of the species monitored, cool season grasses (Carex spp.) were nearly absent, milfoil (Myriophyllum spp.) were relatively unaffected, and pondweed (Potamogeton spp.) increased in biomass and frequency. McGowan et al. (2005) and Hudon (2004) noted similar increases in pondweeds following reductions in lake level.
The frequency of drawdowns may also play a role in determining the species composition of macrophytes. Ruderal (stress-tolerant) or semi-ruderal species may prevail. Ruderal species are characterized as fast growing with early reproduction and high annual seed production (Grime 1977; Rørslett 1989). They tend to have multiple reproduction strategies to increase the likelihood of individual persistence and population growth (Tazik et al. 1982; Siver et al. 1986). Rising water temperatures and concomitant ice-off in the spring promote rapid establishment of ruderal species, limiting growth of other macrophytes (Wagner and Falter 2002).
The viability of seed banks can last for multiple years until suitable germination conditions arise (Howard and Wells 2009). Generally, sediment desiccation stimulates seed germination (Keddy and Reznicek 1986) and facilitates propagation in the drawdown exposure area (McGowan et al. 2005). Species that produce seeds (e.g., Najas minor, Potamogeton pectinatus) are expected to dominate assemblages with increasing lakebed desiccation intensity and frequency (Bornette and Puijalon 2011; Arthaud et al. 2012). Further, the likelihood of persistence for drawdown-tolerant taxa increases because of reduced competition (i.e., for nutrients, light, and space) with more sensitive species (Hellsten 2000; Boschilia et al. 2012).
While drawdowns may favor ruderals, other macrophyte species are restricted to low densities or are extirpated. Taxa most vulnerable in the drawdown exposure zone include perennial species (e.g., many Potamogeton spp.) that rely heavily on rhizomic structures for propagation (Siver et al. 1986), obligate-submergent species (Thomaz et al. 2006; Boschilia et al. 2012), and species sensitive to ice scour (Hellsten 2002).
The response among macrophytes therefore, largely depends on species’ tolerance to freezing, its life-history strategy, and its geographic proximity. Studies summarized from New York (NYSDEC 2005) and Cooke (1980) seem to offer dissimilar results with same plants either positively or negatively affected by drawdown (Tables 2 and 3; e.g., pondweed). Carmignani and Roy (2017), note that assemblage composition is a function of the hydrological components of the drawdown regime (e.g., frequency, duration, and amplitude), competitive interactions, and the species-level tolerance to drawdown-related disturbance (e.g., desiccation, low temperatures, erosional forces) – all factors that likely influence the results documented.
Table 2 – Summary of common macrophytes in New York and their response to winter drawdown.
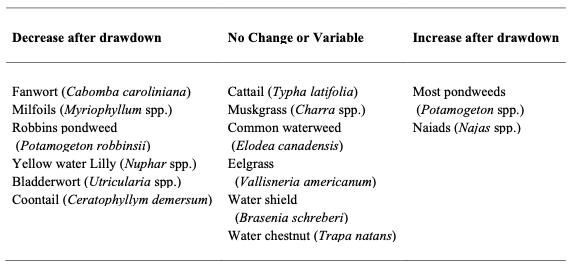
Table 3 – Summary of common macrophytes summarized by Cooke (1980) and their response to winter drawdown.

B. Fish
By reducing the water level, forage fish such as Bluegill, are driven out of shallow water refuges and concentrated in open water, making them more vulnerable to Bass predation. This is a good technique to use in waterbodies that may be overrun with Bluegill because the increased predation by Bass reduces Bluegill numbers and provides additional food for Bass and other predatory fishes. In some cases, routine annual drawdowns have helped managers maintain a balanced Bass/Bluegill fishery. In addition to predatory fish, birds may also benefit by preying on concentrations of fish and invertebrates.
Fredrickson (1991) recommended a slow drawdown to give aquatic organisms an opportunity to disperse from the aquatic plant cover. Brush tops and gravel beds, which serve as fish attractors, can be easily placed while the water is down – thereby providing cover once the lake refills. While studies such as these demonstrate positive effects, removal effects of SAV on fish are somewhat inconsistent, likely because of experimental circumstances and location. For example, Colle and Shireman (1980) report that Bluegill condition and relative abundance declined following water level manipulation (Bettoli et al. 1993). But other research by Cross et al. (1992), Radomski et al. (1995), and Slade (2005) document that Bluegill remain unchanged when vegetation was removed. And still other studies report improvements of survival and population structure (Unmuth et al. 1999). One study performed over a 3-year period demonstrated the resilient nature of fish communities to recover and the ability of aquatic plants to re- sprout after the water body refilled (Paller 2011). So, the effects of water withdrawal on fishes are not definitive.
Shifting plant communities may reduce overhead cover (Bodaly and Lesack 1984) or eliminate spawning grounds previously available to fishes. Water level fluctuations can also alter fish behavior, distribution, and growth. Movement of Largemouth Bass (M. salmoides) and Northern Pike (Esox
lucius), noted by Rogers and Bergersen (1995), occurred in reservoirs between normal and drawdown conditions. Perhaps even more critical to fish is the availability of dissolved oxygen.
Cott et al. (2008), investigated the effects of winter water withdrawal on oxygen concentrations in small (<30 ha) lakes. They demonstrated that oxygen depletion brought on by drawdowns can be compounded by environmental factors like snowfall. Large volumes of water (10 and 20% of their respective under-ice volumes) were withdrawn from two lakes and compared with reference conditions. Under both scenarios, oxygen concentrations were depressed and further reduced with heavier snowpack.
Winter drawdown of reservoirs can induce winterkill if oxygenated water is removed through drawing or flushing the oxygenated surface waters within a reservoir, leaving only poorly oxygenated water (Gaboury and Patalas 1984; Järvalt and Pihu 2002). Drawdowns can also affect fish and other components of the aquatic ecosystem through partial or complete reduction of littoral habitat (refer to habitat section).
C. Macroinvertebrates
Several studies reported significant effects of water level fluctuations on macroinvertebrate abundances and community composition (Furey et al. 2006; Aroviita and Hämäläinen 2008; White et al. 2008, 2011; McEwen and Butler 2010). The amplitude in water level fluctuations or the area exposed by drawdown was a key component to the response among macroinvertebrates who exhibited shorter life cycles and higher mobility in reservoirs with extensive winter drawdowns (Aroviita and Hämäläinen 2008; McEwen and Butler 2010; White et al. 2011).
With respect to taxa richness, research does not provide a decisive result. Aroviita and Hämäläinen (2008) and White et al. (2010, 2011), reported declines in taxa richness among macroinvertebrates sampled at relatively shallow depths (<5 m) but Furey et al. (2006), who sampled areas from 3.5 to 12.5 m reported an overall higher number of taxa in the reservoir, especially below the drawdown zone. These results, much like those reported for fish could, in part, be explained by differences in sampling approaches among studies.
Wetzel (2001) suggests that patchiness in the abundance and distribution of macroinvertebrates within lentic systems can be attributed to species’ physiological limitations, competition, predation, resource availability, and other factors. For example, Rasmussen (1988) showed that littoral zoobenthic biomass was strongly related to submerged macrophyte biomass and localized slope. Both factors can influence the amount of available food as the former provides periphyton and allows for the deposition of organic-rich detritus whereas steep slopes prevent macrophytes from establishing. Because aquatic insects often require emergent vegetation to complete their metamorphosis from larva to adult (Thorp and Covich 1991), winter drawdowns designed to reduce vegetation would directly impact their life cycles.
Winter drawdown can expose bottom substrates to temperatures below 0oC, or lead to ground- fast ice, consequently freezing the associated invertebrate community. This freezing may cause shifts in benthic invertebrate community structure because of species-specific tolerance levels to freezing and temperature regimes. Furey et al. (2006) noted that biomass and densities were greater in a regulated reservoir and that the effects of drawdown on benthic macroinvertebrates extended below the drawdown exposure area. In a similar study, Trottier et al. (2019) report that some species were resistant to desiccation (e.g., Chironomids, Oligochaetes and Sphaeriids), flourishing in the littoral areas of the regulated systems. They caution that such changes could have implications for food web structure and ecosystem function.
D. Other Semi-Aquatic Organisms
Semi-aquatic organisms that are seasonally dependent on aquatic systems for food or refuge (e.g., frogs, turtles, beavers, muskrats, waterbirds) are likely to experience some type of stress or reduction during a portion of their life cycle in association with drawdowns. With respect to birds, aquatic macrophytes offer suitable nest substrate, protective cover, food availability, and territorial requirements (Johnsgard 1956). The physical structure of the vegetation and its successional stage, rather than its taxonomic composition, is of greatest importance to nesting birds (Weller 1978). Hemi- marsh, a 1:1 ratio of emergent vegetation and open water (Weller and Spatcher 1965; Nelson and Bowyer 2000) is commonly recognized as ideal for many bird species throughout later portions of their seasonal cycle. Aquatic vegetation (as foliage, shoots, tubers, and seeds) constitutes a portion of the diet of many birds (Weller 1981). However, invertebrates may represent a greater part of the diet, especially during the breeding period. For example, Murkin et al. (1982) concluded that although blue-winged teal (Anas discors L.) and mallards (Anas platyrhynchos L.) preferred hemi-marsh habitat, their densities were dependent on invertebrate abundance. Similar responses are seen among furbearing mammals.
Muskrats for instance make denning houses in shallow water bodies using mud and aquatic vegetation. Dannel (1978) found that many muskrat lodges were within 1 m of water depth, and the mean water depth at lodge sites was 0.2 m. Relatively constant water levels are required to enable muskrats to access food and construction materials during periods of ice-cover (Glass 1952). Winter water level reductions can prevent semi-aquatic animals such as muskrats from accessing their burrows (Avakyan and Podol’skii 2002).
Water level fluctuation and control is the primary method of muskrat population management as it is the most influential variable determining muskrat abundance, even more so than the type of vegetation present. In winter, muskrats feed on plant material under the ice (Allen and Hoffman 1984); changes to water level regimes that influence the amount of plant material in feeding areas and low water may lead to freezing of food resources that would otherwise be available. Further, when muskrats are forced to search for food during winter, they run an increased risk of being frozen out of their dens (Perry 1982).
If water levels are too low, muskrats are forced to find new feeding areas, increasing their vulnerability to predation and hypothermia. Drought or other low water events can concentrate muskrats, leading to higher rates of social conflict, predation, and disease (Perry 1982). Reduced water levels can prevent the establishment of plants such as cattails (Typha sp.) that muskrats use for food and building materials. Parasite loading and disease also increase with suboptimal water levels (Perry 1982). So, even though a semi-aquatic organism, muskrats could be detrimentally impacted by water withdrawal.
E. Abiotic (Chemical) conditions
Water withdrawals can affect many different aspects of the physical and chemical nature of a waterbody. Water-level manipulation can favor the oxidation and mineralization of organic phosphates (P) (De Groot and Van Wijck 1993). This however largely depends on the precise changes in sediment chemistry, structure, and organic matter sources (e.g., macrophytes, Watts 2000) and the release of sedimentary P on reflooding. For example, Twinch (1987) reported greater releases of P upon- reflooding were a result of changes in sediment composition. Moreover, Klotz and Linn (2001) indicated that P release because of drying was correlated with sediment P and organic content suggesting that drawdowns may increase P loading in lakes rather than decreasing it.
Similarly, sediment desiccation can alter nitrogen (N) cycling by increasing aerobic nitrification and nitrate buildup in sediments (Kadlec 1962) or by mineralizing sedimentary organic-N and intensifying denitrification in underlying anoxic sediments (De Groot and Van Wijck 1993). McGowan et al. (2005) showed that drawdowns of 50% resulted in elevated NH4-N (Ammonia-Nitrate) concentrations but other parameters monitored changed little. Wichelen et al. (2007) report that that drawdown resulted in sediments with a lower water and organic matter content and water column turbidity decreased after the drawdown. But pH in the southern basin of their study declined to <4, probably because sulphides in the sediment were oxidized during drawdown and sediment desiccation (Wichelen et al. 2007). Like biotic variables, the results are varied documenting changes in abiotic factors. Therefore, a good understanding of the chemistry and surrounding geology will be an important consideration before a winter drawdown is considered.
Conclusion
Winter drawdowns can offer many benefits including: reducing SAV and nuisance or overcrowded fish populations, changing the composition of plant and animal species, and providing electrical power in winter and water storage in spring. However, the timing and duration of drawdowns can negatively impact species by compressing life histories and changing food web structure.
Regarding Deep Creek Lake, data should continue to be collected concerning the spread and or invasion of various plant species like those listed in Table 1. Identification of all aquatic plants will be an important part of this process as this may determine the efficacy and longevity of drawdowns. In addition, available fish data should be evaluated to determine the species composition and if possible, the size structure of various populations; moreover, other data from the lake should be incorporated as available (e.g., bird or macroinvertebrate surveys and chemical information). A timeline similar to that proposed by WDFW (2015; e.g., Table 2) should be developed to achieve the desired goals of the Deep Creek Watershed Foundation.
The proposed dredging of Arrowhead Run Cove may prove instructive to assess the physical changes associated with drawdowns and the mechanical removal of both plants and silt. This could serve as a model of sorts to determine what other data should be assessed to determine the efficacy of such an action at Deep Creek Lake and what the implications may be.
Literature Cited
Aiken, S. G., P. R. Newroth, and I. Wile. 1979. The biology of Canadian weeds. Myriophyllum spicatum L. Canadian Journal of Plant Science 59: 201-215.
Allen, A.W. and R.D. Hoffman. 1984. Habitat Suitability Index Models: Muskrat. U.S. Fish and Wildlife Service, FWS ⁄ OBS82 ⁄ 10.46. 27 pp.
Aroviita, J. and H. Hämäläinen. 2008. The impact of water-level regulation on littoral macroinvertebrate assemblages in boreal lakes. Hydrobiologia 613:45-56.
Arthaud, F., D. Vallod, J. Robin, and G. Bornette. 2012. Eutrophication and drought disturbance shape functional diversity and life history traits of aquatic plants in shallow lakes. Aquatic Science 74:471- 481.
Avakyan, A.B. and S.A. Podol’skii. 2002. Impact of Reservoirs on the Fauna. Water Resources 29(2):123-132.
Beklioglu, M., G. Altinayar, and C.O. Tan. 2006. Water level control over submerged macrophyte development in five shallow lakes of Mediterranean Turkey. Archiv für Hydrobiologie 166:535-556.
Bettoli, P.W., M.J. Maceina, R.L. Noble, and R.K. Betsill. 1993. Response of a reservoir fish community to aquatic vegetation removal. North American Journal of Fisheries Management 13:110-124.
Bodaly, R.A. and L.F.W. Lesack. 1984. Response of a Boreal Northern Pike (Esox lucius) Population to Lake Impoundment: Wupaw Bay, Southern Indian Lake, Manitoba. Canadian Technical Report of Fisheries and Aquatic Sciences 41:706-714.
Bornette G. and S. Puijalon. 2011. Response of aquatic plants to abiotic factors: a review. Aquatic Science 73:1-14.
Boschilia S.M., E.F. De Oliveira, and A. Schwarzbold. 2012. The immediate and long-term effects of water drawdown on macrophyte assemblages in a large subtropical reservoir. Freshwater Biology 57:2641-2651.
Carmignani, J.R. and A.H. Roy. 2017. Ecological impacts of winter water level drawdowns on lake littoral zones: a review. Aquatic Sciences 79:803-824.
Colle, D.E. and J.V. Shireman. 1980. Coefficients of condition for largemouth bass, bluegill, and redear sunfish in Hydrilla-infested lakes. Transactions of the American Fisheries Society 109:521-531.
Cooke, G.D. 1980. Lake level drawdown as a macrophyte control technique. Journal of the American Water Resources Association 16:317-322.
Cooke, G.D., E.B Welch, S.A Peterson, and S.A. Nichols. 2005. Restoration and management of lakes and reservoirs, 3rd ed. CRC Press Boca Raton, Florida.
Cott, P.A., P.K. Sibley, A.M. Gordon, R.A. Bodaly, K.H. Mills, W.M. Somers, and G.A. Fillatre. 2008. The Effects of Water Withdrawal from Ice-Covered Lakes on Oxygen, Temperature and Fish. Journal of the American Water Resources Association 41:328-342.
Cross, T.K., M.C. McInerny, and R.A. Davis. 1992. Macrophyte removal to enhance bluegill, largemouth bass, and northern pike populations. Minnesota Department of Natural Resources, Section of Fisheries Investigational Report 415, St. Paul, Minnesota.
Dannel, K. 1978. Intra- and Interannual Changes in Habitat Selection by the Muskrat. Journal of Wildlife Management 42(3):540- 549.
Cooley, P.M. and W.G. Franzin. 2008. Predicting the spatial mud energy and mud deposition boundary depth in a small boreal reservoir before and after draw down. Lake and Reservoir Management 24: 261-272.
Craig, V.E. 1952. A story of fish production as it applies to Montana. M.S. Thesis. Montana State University, Bozeman, Montana.
Dvorak, J. and E.P. Best. 1982. Macro-invertebrate communities associated with the macrophytes of Lake Vechten: structural and functional relationships. Hydrobiologia 95:115-126.
Effler, S.W. and D.A. Matthews. 2004. Sediment resuspension and drawdown in a water supply reservoir. Journal of the American Water Resources Association 40: 251-264.
Eiswerth, M.E., S.G. Donaldson, and W.S. Johnson. 2000. Potential environmental impacts and economic damages of Eurasian watermilfoil (Myriophyllum spicatum) in western Nevada and
northeastern California. Weed Technology 14(3):511-518.
Fox J., P.L. Brezonick, and M.A Keirn. 1977. Lake drawdown as a method for improving water quality. U.S. Environmental Protection Agency, Corvallis Environmental Research Laboratory, Corvallis, Oregon.
Fredrickson, L.H. 1991. Strategies for water level manipulation in moist-soil systems. U.S. Fish and Wildlife Service. Leaflet 13.4.6. 8 pp.
Furey, P.C., R.N. Nordin, and A. Mazumder. 2004. Water level drawdown affects physical and biogeochemical properties of littoral sediments of a reservoir and a natural lake. Lake and Reservoir Management 20: 280-295.
Furey, P.C., R.N. Nordin, and A. Mazumder. 2006. Littoral Benthic Macroinvertebrates Under Contrasting Drawdown in a Reservoir and a Natural Lake. Journal of the North American Benthological Society 25(1):19-31.
Gaboury, M.N. and J.W. Patalas. 1984. Influence of Water Level Drawdown on the Fish Populations of Cross Lake, Manitoba. Canadian Journal of Fisheries and Aquatic Sciences 41:118-125.
Glass, B.P. 1952. Factors Affecting the Survival of the Plains Muskrat Ondatra Zibethica Cinnamomina in Oklahoma. Journal of Wildlife Management 16:484-491.
Grime, J. 1977. Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. American Naturalist 111:1169-1194.
Groen, C.L. and T.A. Schroeder. 1978. Effects of water level management on walleye and other coolwater fishes in Kansas reservoirs. American Fisheries Society Special Publication 11:278–283.
Hellsten, S.K. 1997. Environmental factors related to water level regulation-a comparative study in northern Finland. Boreal Environmental Research 2:345-367.
Hellsten, S.K. 2000. Environmental factors and aquatic macrophytes in the littoral zone of regulated lakes: causes, consequences and possibilities to alleviate harmful effects. Thesis, University of Oulu, Oulu, Finland.
Howard R.J. and C.J. Wells. 2009. Plant community establishment following drawdown of a reservoir in southern Arkansas, USA. Wetlands Ecological Management 17:565-583.
Hudon, C. 2004. Shift in Wetland Plant Composition and Biomass Following Low-Level Episodes in the St. Lawrence River Aquatic Vegetation: Looking Into the Future. Canadian Journal of Fisheries and Aquatic Sciences 61:603-617.
Hulsey, A.H. 1957. Effects of a fall and winter drawdown on a flood control lake. In: Proceedings of the annual conference southeastern association of game and fish commissioners 10:285-289.
James, W.F., J.W. Barko, H.L. Eakin, and D.R. Helsen. 2001. Changes in sediment characteristics following drawdown of Big Muskego Lake, Wisconsin. Archiv Fur Hydrobiologie 151:459-474.
Järvalt, A. and E. Pihu. 2002. Influence of Water Level on Fish Stocks and Catches in Lake Võrtsjärv. Proceedings of the Estonian Academy of Sciences. Biology and Ecology 51:74-84.
Johnsgard, P.A. 1956. Effects of water fluctuation and vegetation change on bird populations, particularly waterfowl. Ecology 37: 689-701.
Johnstone, I.M., B.T. Coffey, and C. Howard-Williams. 1985. The role of recreational boat traffic in interlake dispersal of macrophytes: a New Zealand case study. Journal of Environmental Management 20:263-279.
Keddy, P.A. and A.A. Reznicek. 1986. Great Lakes vegetation dynamics: the role of fluctuating water levels and buried seeds. Journal of Great Lakes Research 12:25-36.
Keto, A., A. Tarvainen, and S. Hellsten. 2006. The effect of water level regulation on species richness and abundance of aquatic macrophytes in Finnish lakes. Verhandlungen des Internationalen Verein Limnologie 29:2103-2108.
Klotz, R.L. and S.A. Linn. 2001. Influence of factors associated with water level drawdown on phosphorus release from sediments. Lake and Reservoir Management 17:48-54.
Korschgen, C.E. and W.L. Green. 1988. American wild celery (Vallisneria americana): ecological considerations for restoration (No. 19). US Department of the Interior, Fish and Wildlife Service.
Lantz, K.E., J.T. Davis, J.S. Hughes, and H.E. Schafer Jr. 1967. Water level fluctuation-its effects on vegetation control and fish population management. In: Proceedings of the Annual Conference Southeastern Association of Game and Fish Commissioners 18:483-494.
Madsen, J.D., L.W. Eichler, and C.W. Boylen. 1988. Vegetative spread of Eurasian watermilfoil in Lake George, New York. Journal of Aquatic Plant Management 26:47-50.
McEwen, D.C. and M.G. Butler. 2010. The effects of water-level manipulation on the benthic invertebrates of a managed reservoir. Freshwater Biology 55:1086-1101.
McGowan, S., P.R. Leavitt, and R.I. Hall. 2005. A Whole-Lake Experiment to Determine the Effects of Winter Drought on Shallow Lakes. Ecosystems 8(6):694-708.
Minnesota DNR (MN DNR). 2022. Wild celery (Vallisneria americana). .
Mississippi Interstate Cooperative Resource Association (MICRA). 2015. The use of grass carp in the United States. A report to the US Fish and Wildlife Service – Agreement #F12AP00630. .
Mjelde, M., S. Hellsten, and F. Ecke. 2012. A water level drawdown index for aquatic macrophytes in Nordic lakes. Hydrobiologia 704:141-151.
Murkin, H. R., R. M. Kaminski, and R. D. Titman. 1982. Responses of dabbling ducks and aquatic invertebrates to an experimentally manipulated cattail marsh. Canadian Journal of Zoology 60: 2324-2332.
Nelson, T. and M. Bowyer. 2000. Managing wetland vegetation for marshbirds and waterfowl. Final project report: FY00 Wildlife Preservation Fund, Eastern Illinois University, Charleston, Illinois.
New York State DEC (NYSDEC). 2005. A primer on aquatic plant management in New York State. NYSDEC Division of Water. Draft Report.
Nichols, S.A. and B.H. Shaw. 1986. Ecological life histories of the three aquatic nuisance plants, Myriophyllum spicatum, Potamogeton crispus, and Elodea canadensis. Hydrobiologia 131:3-21.
Paller, M.H. 2011. Recovery of a reservoir fish community from drawdown related impacts. North American Journal of Fisheries Management 17:726-733.
Perry, R. 1982. Muskrats. In: Wild Mammals of North America, J. Chapman and G. Feldhamer (Editors). John Hopkins University Press, Baltimore, Maryland, 1147 pp.
Radomski, P.J., T.J. Goeman, and P.D. Spencer. 1995. The effects of chemical control of submerged vegetation on the fish community of a small Minnesota centrarchid lake. Minnesota Department of Natural Resources, Section of Fisheries Investigational Report 442, St. Paul, Minnesota.
Reznicek, A.A., E.G. Voss, and B.S. Walters. 2011. Michigan Flora Online. University of Michigan.
Rogers, K.B. and E.P. Bergersen. 1995. Effects of a Fall Drawdown on Movement of Adult Northern Pike and Largemouth Bass. North American Journal of Fisheries Management 15:596-600.
Rørslett, B. 1989. An integrated approach to hydropower impact assessment. II. Submerged macrophytes in some Norwegian hydro-electric lakes. Hydrobiologia 175:65-82.
Siver, P.A., A.M. Coleman, G.A. Benson, and J.T. Simpson. 1986. The effects of winter drawdown on macrophytes in Candlewood Lake, Connecticut. Lake Reservoir Management 2:69-73.
Slade, J.G. 2005. Evaluation of fish-habitat relationships following a species-specific herbicide treatment in Minnesota. M.S. thesis, Mississippi State University, Mississippi State.
Smith C.G. and J.W. Barko. 1990. Ecology of Eurasian watermilfoil. Journal of Aquatic Plant Management 28:55-64.
Sutela T., J. Aroviita, and A. Keto. 2013. Assessing ecological status of regulated lakes with littoral macrophyte, macroinvertebrate and fish assemblages. Ecological Indicators 24:185-192.
Tarver, D.P. 1980. Water fluctuation and the aquatic flora of Lake Miccosukee. Journal of Aquatic Plant Management 18:19-23.
Tazik, P.P., W.R. Kodrich, and J.R. Moore. 1982. Effect of overwinter drawdown on bushy pondweed. Journal of Aquatic Plant Management 20:19-21.
Texas A&M. 2022a. How to control eelgrass: A diagnosis tool for pond plants and algae. .
Texas A&M. 2022b. How to control large-leafed pondweed: A diagnosis tool for pond plants and algae. .
Thomaz, S.M., T.A. Pagioro, L.M. Bini, and K.J Murphy. 2006. Effect of reservoir drawdown on biomass of three species of aquatic macrophytes in a large sub-tropical reservoir (Itaipu, Brazil). Hydrobiologia 570:53-59.
Thorp, J.H. and A.P. Covich, 1991. Ecology and Classification of North American Freshwater Invertebrates. Academic Press, San Diego, California, 911 pp.
Tomcko, C.M. and D.L. Pereira. 2006. The relationship of bluegill population dynamics and submerged aquatic vegetation in Minnesota lakes. Minnesota Department of Natural Resources, Investigative Report 538.
Trottier, G., H. Embke, K. Turgeon, C. Solomon, C. Nozais, and I. Gregory-Eaves. 2019. Macroinvertebrate abundance is lower in temperate reservoirs with higher winter drawdown. Hydrobiologia 834:199-211.
Turner, M.A., D.B. Huebert, D.L. Findlay, L.L. Hendzel, W.A. Jansen, R.A. Bodaly, L.M. Armstrong, and S.E.M. Kasian. 2005. Divergent Impacts of Experimental Lake-Level Drawdown on Planktonic and Benthic Plant Communities in a Boreal Forest Lake. Canadian Journal of Fisheries and Aquatic Sciences 62:991-1003.
Twinch, A.J. 1987. Phosphate exchange characteristics of wet and dried sediments samples from a hypertrophic reservoir. Implications for the measurement of phosphorus states. Water Resources 21:1225-1230.
University of Florida IFAS Center for Aquatic and Invasive Plants. 2021. Sections 3. Control Methods. .
Verrill, D.D. and C.R. Berry Jr. 1995. Effectiveness of an electric barrier and lake drawdown for reducing common carp and bigmouth buffalo abundances. North American Journal of Fisheries Management 15:137-141.
Voss, E.G. and A.A. Reznicek. 2012. Field manual of Michigan flora. University of Michigan Press, Ann Arbor, Michigan.
Wagner, T. and C.M. Falter. 2002. Response of an aquatic macrophyte community to fluctuating water levels in an oligotrophic lake. Lake Reservoir Management 18:52-65.
Wakeman, R.W. and D.H. Les. 1994. Optimum growth conditions for Potamogeton amplifolius, Myriophyllum spicatum and Potamogeton richardsonii. Lake and Reservoir Management 9:129-133.
Washington Department of Fish and Wildlife (WDFW). 2015. Aquatic plants and fish: rules for aquatic plant removal and control. Washington Department of Fish and Wildlife.
Weller, M.W. 1978. Management of freshwater marshes for wildlife. p. 267-284. In R. E. Good, D. F.
Whigham, and R. L. Simpson (eds.) Freshwater Wetlands, Ecological Processes and Management Potential. Academic Press, New York, NY, USA.
Weller, M.W. 1981. freshwater Marshes. University of Minnesota Press, Minneapolis, MK USA.
Weller, M.W. and C.S. Spatcher. 1965. Role of habitat in the distribution and abundance of marsh birds. Iowa Agricultural and Home Economics Experiment Station. Ames, IA. Special Report No. 43.
Wetzel, R.G. 2001. Limnology: Lake and River Ecosystems. Gulf Professional Publishing, Houston.
White, M.S., M.A. Xenopoulos, K. Hogsden, R.A Metcalfe, and P.J. Dillon. 2008. Natural level fluctuation and associated concordance with water quality and aquatic communities within small lakes of the Laurentian Great Lakes region. Hydrobiologia 613:21-31.
White, M.S., M.A. Xenopoulos, R.A. Metcalfe, and K.M. Somers. 2010. On the role of natural water level fluctuation in structuring littoral benthic macroinvertebrate community composition in lakes. Limnology and Oceanography 55:2275-2284.
White, M.S., M.A. Xenopoulos, R.A. Metcalfe, and K.M. Somers. 2011. Water level thresholds of benthic macroinvertebrate richness, structure, and function of boreal lake stony littoral habitats. Canadian Journal of Fisheries and Aquatic Sciences 68:1695-1704.
Wichelen, J.V., S. Declerck, K. Muylaert, I. Hoste, V. Geenens, J. Vanderkerkhove, E. Michels, N. DePauw, M. Hoffmann, L. DeMeester, and W. Vyverman, 2007. The importance of drawdown and sediment removal for the restoration of the eutrophied shallow Lake Kraenepoel (Belgium) Hydrobiologia 584:291-303.
Wilcox, D.A. and J.E. Meeker. 1991. Disturbance effects on aquatic vegetation in regulated and unregulated lakes in northern Minnesota. Canadian Journal of Botany 69: 1542-1551.

